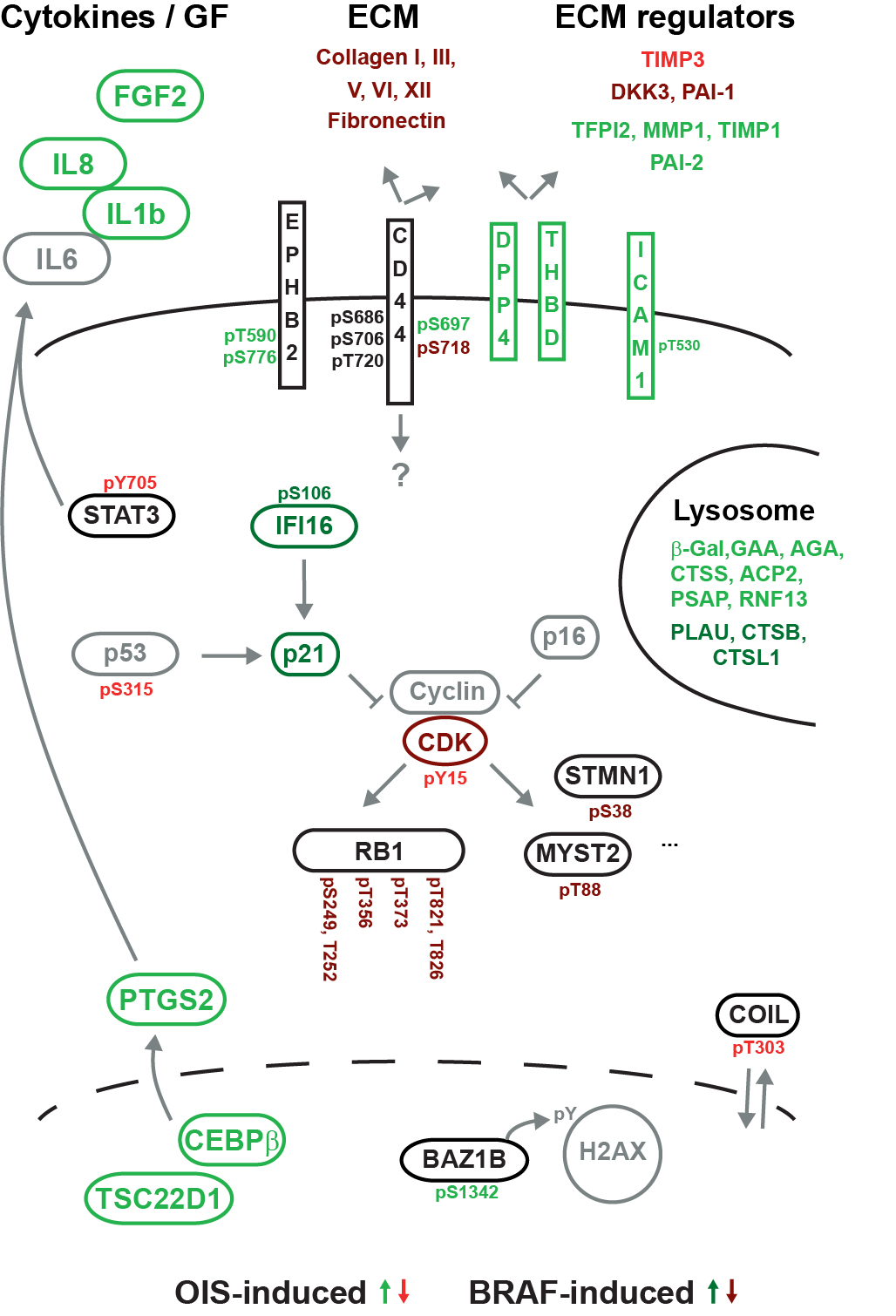
Expression of the BRAF(V600E) oncoprotein is known to cause benign lesions, such as melanocytic nevi (moles). Despite the oncogenic function of mutant BRAF, these lesions are arrested by a cell-autonomous mechanism called oncogene-induced senescence. Infrequently, nevi can progress to malignant melanoma, through mechanisms that are incompletely understood. To gain more insight into this vital tumor-suppression mechanism, we performed a mass-spectrometry-based screening of the proteome and phosphoproteome in cycling and senescent cells and in cells with abrogated senescence. Proteome analysis of senescent cells revealed the up-regulation of established senescence biomarkers, including specific cytokines, but also several proteins not previously associated with senescence, including extracellular matrix-interacting. Using both general and targeted phosphopeptide enrichment by Ti(4+)-IMAC and phosphotyrosine antibody enrichment, we identified over 15,000 phosphorylation sites. Among the regulated phosphorylation sites we encountered components of the interleukin, BRAF/MAPK, and CDK-retinoblastoma pathways and several other factors. The extensive proteome and phosphoproteome dataset of BRAF(V600E)-expressing senescent cells provides molecular clues as to how oncogene-induced senescence is initiated, maintained, or evaded, serving as a comprehensive proteomic basis for functional validation.
de Graaf EL, Kaplon J, Zhou H, Heck AJ, Peeper DS, Altelaar AF.
Mol Cell Proteomics. 2014 Aug;13(8):2089-2100.
![]()